|
This finding revolutionized the way scientists thought about the atom and had major ramifications for the field of physics. Thomson announced his discovery that atoms were made up of smaller components. On April 30, 1897, British physicist J.J. 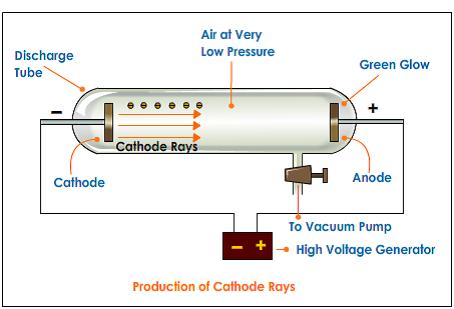
When did JJ Thomson discovered the atomic theory? The positive particle was named the proton. This resulted in The positive particle had a charge equal and opposite to the electron. 
What was Goldstein experiment?Įugene Goldstein discovered positive particles by using a tube filled with hydrogen gas (this tube was similar to Thomson’s tube). Niels Bohr built upon Rutherford’s model to make his own. The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre. In his classic experiment, Thomson measured the mass-to-charge ratio of the cathode rays by measuring how much they were deflected by a magnetic field and comparing this with the electric deflection. These negatively charged particles are deflected from their straight-line path when an electric field is applied. Thomson discovered that cathode ray particles carry a negative charge. Thomson’s cathode ray experiments determine quizlet? A cathode ray tube is made of glass with a small amount of some kind of gas in it. Why did Thomson’s results from experimenting with cathode rays cause a big change in scientific thought about atoms? His results gave the first evidence that atoms were made up of smaller particles. Why did Thomson’s results from experimenting with cathode rays cause a big change in scientific thought about atoms? The presence of cathode rays were confirmed by making a hole in the anode which resulted in cathode rays striking the fluorescent zinc sulphide (ZnS) coating on the tube behind the anode. Every particle who is moving with some velocity, has momentum. When cathode rays rotate a light paddle wheel placed in his path then we conclude that the ray have some mass (mass of electrons). What conclusion is made from the observation that cathode rays rotate a paddle wheel placed in their path? When they strike the light paddle wheels blades the momentum is shared and it starts rotating. Because these particles move with a velocity. What happens when a very light paddle wheel is placed in the path of cathode rays?Ī light paddle will rotate when it is placed in the path of α,βor cathod rays. He also found the first evidence that stable elements can exist as isotopes and invented one of the most powerful tools in analytical chemistry – the mass spectrometer. Thomson took science to new heights with his 1897 discovery of the electron – the first subatomic particle. Why was JJ Thomson’s discovery important? He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. What experiment did JJ Thomson used to discover the electron?ĭevelopment of the Atomic Theory. 
They were first observed in Crookes tubes during experiments by the German scientist Eugen Goldstein, in 1886. How was anode rays discovered?Īn anode ray (also positive ray or canal ray) is a beam of positive ions that is created by certain types of gas-discharge tubes. He found that when the rays entered the slit in the cylinders, the electrometer measured a large amount of negative charge. Thomson wanted to see if, by bending the rays with a magnet, he could separate the charge from the rays. Why did Thomson apply a magnetic field in his experiment? Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” What does the paddle wheel experiment show?Ĭrookes attributed the motion of the wheel to momentum transfer from the cathode rays (electrons) to the wheel, and interpreted the experiment as providing evidence that cathode rays were particles. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
